Certificate
ISO CERTIFICATE
Why ISO so important?
ISO 13485 ensures that manufacturers continue to design, develop, produce, install and deliver safely and in compliance with relevant regulatory requirements and the intended purpose of medical devices.
ISO 13485:2016 is the standard for a Quality Management System (“QMS”) for the design and manufacture of Medical Devices.
GDPMD CERTIFICATE
Why GDPMD so important?
The objective of GDPMD is to ensure the quality, safety and performance of medical devices which include but are not limited to product sourcing and procurement; transportation and delivery; storage; installation, commissioning, service and maintenance, calibration and after sales service; tracking, documentation and record-keeping practices.
MDA CERTIFICATE
Why MDA so important?
For the purpose of placing a medical device in the market: the manufacturer, Authorised Representative of foreign manufacturer, importer and distributor shall establish, maintain and implement appropriate quality management system that is commensurate with the role and function of the establishment.
Under the Medical Device Act (Act 737) and the Medical Device Authority Act 2012 (Act 738) all medical devices manufactured, imported, or distributed in Malaysia require a registration.
The Medical Device Authority (MDA) is a division of the Ministry of Health Malaysia (MOH) in charge of regulating medical device and its industry players in Malaysia.
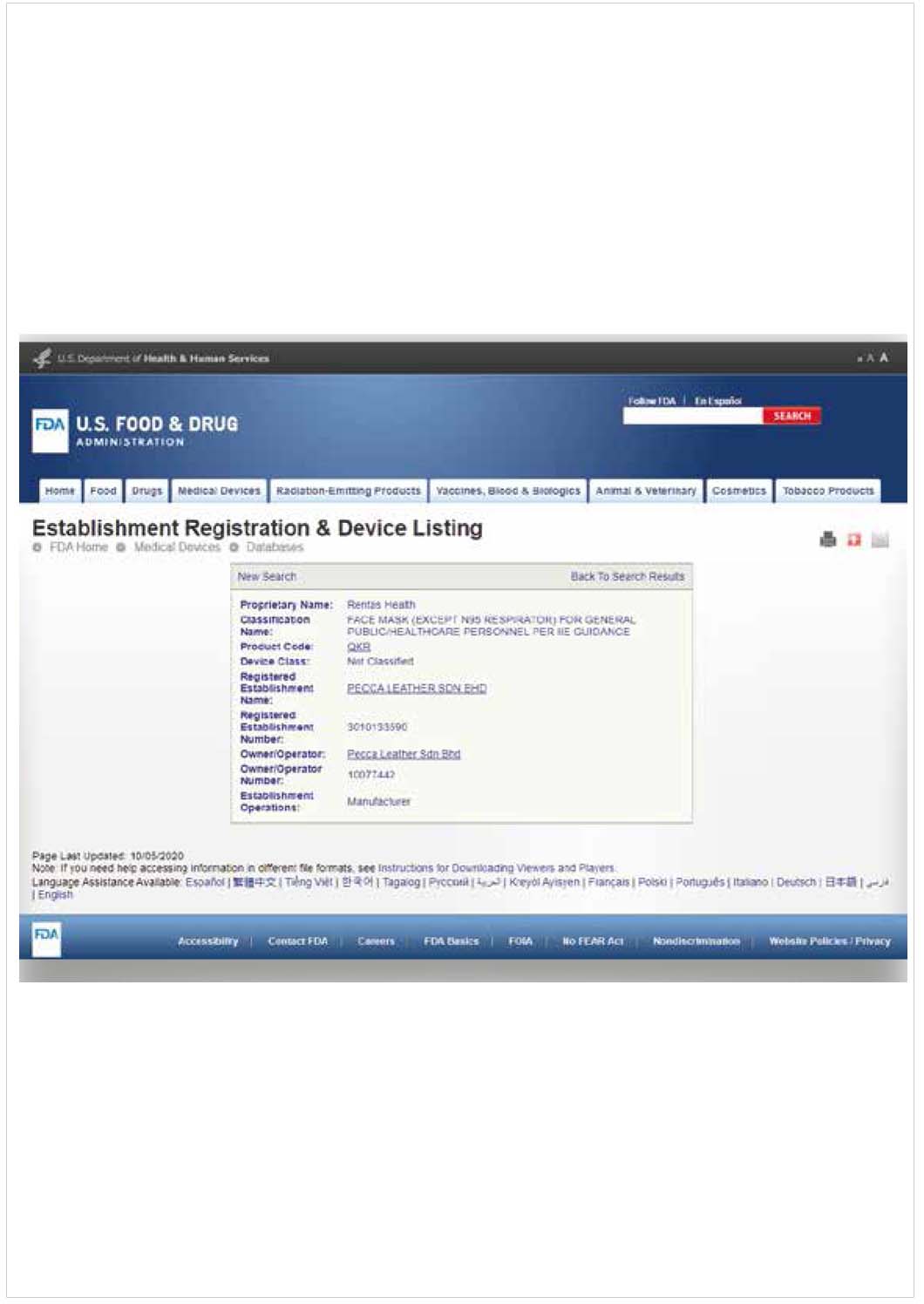
FDA CERTIFICATE
Why FDA so important?
The Food and Drug Administration is responsible for advancing the public health by helping to speed innovations that make medical products more effective, safer, and more affordable and by helping the public get the accurate, science-based information they need to use medical products and foods to maintain and improve their health.
CE CERTIFICATE
Why CE so important?
CE marking is an important procedure that guarantees a product’s conformity to EU regulations. Manufacturers, importers and distributors of non-food products are obliged to provide the CE marking if they want to trade in EU/EAA markets.
Selling products without the CE mark is an economic offence that is punished differently by each Member State of the European Union. Affixing CE marking on products that do not require the CE can also lead to penalties.
FILTRATION EFFICIENCY TESTING (BFE & PFE)
Filtration Efficiency Testing (BFE & PFE)
Bacterial Filtration Efficiency and Particle Filtration Efficiency are probably the most important tests to be conducted on medical textile products such as surgical masks, gowns or caps, as well as on air filters.
SIRIM CERTIFICATE
Why SIRIM so important?
SIRIM (Standard & Industrial Research Institute of Malaysia) is the national organisation for standards and quality and as a promoter of technological excellence in the Malaysian industry.
MADE IN MALAYSIA CERTIFICATE
Why Made In Malaysia so important?
Ministry of Domestic Trade and Consumer Affairs (KPDNHEP) is responsible for processing application and issuing Made in Malaysia Logo marking to private or partnership businesses, private limited companies and cooperatives upon compliance with the requirements.
The Made in Malaysia Logo marking is an endorsement that the product is manufactured in Malaysia (country of origin).